
Food Safety Assessment Methodology - Revision 6
FSIS directive
5100.1
Series Type
5000 Series: Program Services
Issue Date
Dec 17, 2024
Full Directive
CHAPTER I - GENERAL
- PURPOSE
This directive instructs Enforcement, Investigations and Analysis Officers (EIAOs) and EIAO-trained Public Health Veterinarians (PHVs) on how to conduct Food Safety Assessments (FSAs) within seven production days. This directive also provides instructions on how to document FSAs using the FSA tools in the FSA Module in the Public Health Information System (PHIS). The FSA tools are a series of questionnaires that guide the EIAO in gathering information to assess the establishment's food safety system. FSIS is reissuing this directive to include instructions for Frontline Supervisors (FLS) to conduct an in-person follow-up visit when the EIAO identifies systemic issues during the FSA that do not rise to the level of an enforcement action. FSIS has also updated links to the latest guidance in this directive.
NOTE: In this directive, the term “EIAO” also refers to EIAO-trained PHVs when they are conducting EIAO activities. The term “District Office (DO)” includes the District Manager, the Deputy District Manager, the Supervisory Enforcement Investigations and Analysis Officer (SEIAO), and the District Case Specialist (DCS).
KEY POINTS- Through the FSA, EIAOs verify whether Hazard Analysis and Critical Control Point (HACCP) systems are validated and implemented effectively through records review and observation;
- EIAOs ensure responses in the FSA tools support the recommended outcome; and
- EIAOs communicate with establishment management, inspection program personnel (IPP), the frontline supervisor (FLS), and the DO throughout the entire FSA.
- CANCELLATION
FSIS Directive 5100.1, Revision 5, Food Safety Assessment Methodology, 10/19/22. - BACKGROUND
- The FSA is an assessment of an establishment's food safety system to verify that meat, poultry, or egg products are safe, wholesome, and produced in accordance with FSIS statutory and regulatory requirements (i.e., the Acts, specifically the Federal Meat Inspection Act, the Poultry Products Inspection Act, and the Egg Products Inspection Act, and 9 CFR).
- The FSA tools provide the EIAO with a structured framework for conducting and documenting the FSA. The FSA tools consist of a General tool and several commodity-specific tools. Table 1 provides a list of product types, HACCP categories, and the applicable commodity-specific FSA tool.
- FSA OVERVIEW
- FSAs are performed when the DO determines that an FSA is appropriate based on its analysis of the Public Health Risk Evaluation (PHRE), as described in FSIS Directive 5100.4, Public Health Risk Evaluation (PHRE) Methodology.
- The EIAO is to complete the in-plant portion of the FSA within seven production days. Production days are the days the establishment is producing the product relevant to the FSA. The FSA may be extended if additional time is necessary to develop the recommendation for an enforcement action, or if three or more tools are selected for completion during the FSA (e.g., a new establishment coming under a grant of inspection).
- The EIAO is to use FSA tools to record findings and to determine whether:
- The HACCP system is designed to prevent, reduce, or eliminate the hazards identified in the hazard analysis;
- The establishment's decisions in its hazard analysis are appropriately supported, including by the establishment's validation documents; and
- The establishment's sampling programs are designed appropriately and performed using validated testing methods that are fit for purpose, and the establishment reacts appropriately to sampling results.
- The EIAO is to analyze the answers to the questions in the FSA tools to reach a logical and supportable recommendation that:
- No action is necessary;
- The in-plant team is to issue a noncompliance record (NR) for any regulatory noncompliance; or
- The DO is to issue an enforcement action, either a Notice of Intended Enforcement (NOIE) with or without NRs or a Notice of Suspension (NOS) (FSIS Directive 5100.3, Administrative Enforcement Action Decision-Making and Methodology).
- In responding to questions in the tools, the EIAO is to focus on documenting noncompliance and vulnerabilities, not making positive editorial findings. At the end of each tool, the EIAO is to summarize the findings that bear directly on their recommendation. The recommendation is to be supported by FSIS statutory and regulatory requirements (i.e., the Acts and 9 CFR).
NOTE: EIAOs are to be aware that a vulnerability is a weakness in the establishment's process that does not rise to the level of noncompliance but that could negatively contribute to the establishment's ability to produce safe and wholesome meat, poultry, or egg products. - The EIAO Process Overview and FSA Scheduling Workflow diagrams shown below in Figure 1 and Figure 2 provide a visual depiction of the FSA process.
Figure 1. EIAO Process Overview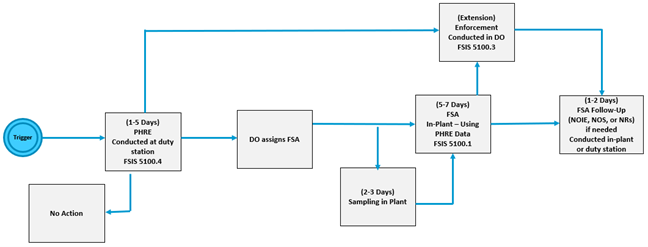
Figure 2. FSA Scheduling Workflow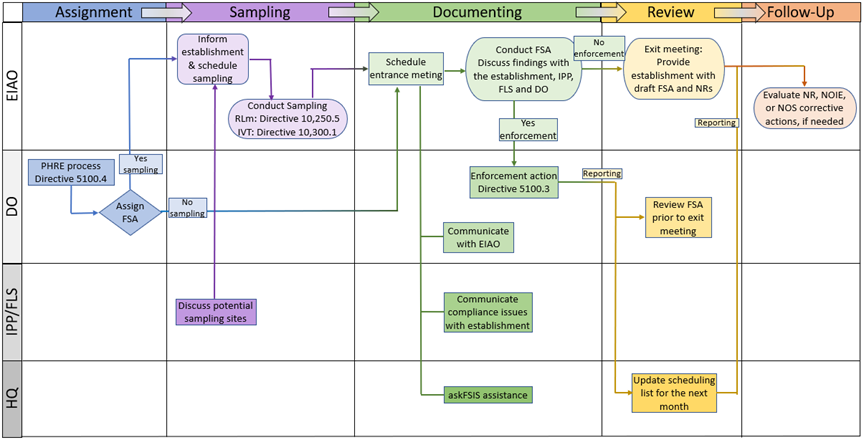
- GENERAL FSA METHODOLOGY
- The EIAO is to evaluate the HACCP system as a whole. The HACCP system includes the hazard analysis, any supporting documentation, including prerequisite programs, supporting decisions in the hazard analysis, and all HACCP records. Therefore, the EIAO is to consider all documentation that affects decisions in the hazard analysis when developing a recommendation.
- The EIAO is not to only verify whether the establishment meets individual regulatory requirements but is also to focus on the vulnerabilities that may affect the food safety system and the establishment's ability to produce safe and wholesome meat, poultry, or egg products.
- In general, the EIAO is to conduct the assessment by:
- Directly observing establishment implementation as described in section VII. At a minimum, the EIAO is to observe the establishment carrying out its HACCP verification procedures, Sanitation Standard Operating Procedures (Sanitation SOPs), and sampling and evaluation of test results; and
- Reviewing a random selection of 13 days of records and documentation specific to the HACCP plan targeted (see chapter II, section VIII).
- The EIAO is to use the relevant FSIS documents, including this directive and the directives and guidelines referenced in chapter II, section VI and section VII, to evaluate the establishment's HACCP system.
NOTE: The EIAO is to be aware that guidelines represent FSIS' best practice recommendations and do not represent requirements that must be met. Establishments may choose to adopt different procedures than those outlined in a guideline, but they would need to support why those procedures are effective.
- COMMUNICATION
- The EIAO is to request information from the establishment, not demand it, and explain to establishment officials FSIS' statutory authority under the Acts to examine facilities and to copy records. If the establishment is unwilling to provide information, the EIAO is to communicate with the DO to develop a strategy for gaining access to necessary information. The DO is to contact the Executive Associate for Regulatory Operations to coordinate the involvement of the Office of Investigation, Enforcement, and Audit and the issuance of an administrative subpoena to obtain such records, if necessary, because the establishment continues to deny access to the facilities and to copy records.
- Requests and exchange of records, in-progress updates, and other information pertaining to the FSA should be performed as efficiently as possible to support both the EIAO and establishment time resource management.
- The EIAO is to describe any identified noncompliance or vulnerabilities with the establishment as soon as possible after identification. The EIAO is not to predict the possible FSA outcomes during the FSA.
- An establishment's attempt to bring itself into compliance upon being notified of a noncompliance finding during the FSA does not negate the noncompliance finding. IPP are to document noncompliance in NRs; the EIAO may assist IPP by providing the narrative for the NR. The EIAO is to document descriptions of noncompliance in the FSA. If the EIAO recommends an enforcement action to the DO, the EIAO is to document relevant noncompliances in the NOIE or NOS.
- The EIAO is to discuss their findings and recommendations with the DO to ensure that all scientific, technical, and regulatory or statutory issues in the EIAO's report have been resolved. The DO may request additional information from the EIAO or may provide additional resources.
- The EIAO is to communicate with the IPP and FLS throughout the course of the assessment.
- The EIAO, IPP, and FLS are to work collaboratively to ensure that all noncompliances are documented on NRs and communicated to establishment management either during the FSA or at the exit meeting. The EIAO is to notify the FLS, IPP, and the establishment management immediately when the EIAO observes a noncompliance that has an immediate impact on food safety. Other noncompliances, such as design or recordkeeping issues, should be presented at the exit meeting. The EIAO is to document all noncompliances identified during the FSA in the relevant tools, irrespective of whether the NRs were provided to the establishment at the exit meeting or earlier.
- The EIAO is to provide frequent updates to the IPP and FLS to inform them of the findings and of any recommendations that the EIAO is planning to make.
CHAPTER II - FSA
- FSA PREPARATION
- When the DO decides that an FSA is necessary based on the PHRE, the EIAO is to document the assessment plan, as described in FSIS Directive 5100.4.
- The EIAO is to determine which tools will be completed based on the areas of concern (see Table 1). The EIAO is to complete the General tool for every FSA and for most FSAs, one other tool (e.g., Meat and Poultry tool for Shiga toxin-producing Escherichia coli (STEC) positives, or Ready-To-Eat (RTE) Processed Products tool for Lm positives or RLm sampling). The following are situations where the EIAO could complete more than two tools:
- In new establishments coming under a grant of inspection, all tools covering the HACCP categories for products the establishment produces are to be selected. The EIAO is to focus on initial validation. Section IX provides instructions for verifying an establishment's scientific support and in-plant validation data;
- Criteria in FSIS Directive 5100.4 span multiple HACCP categories (e.g., STEC positive in raw non-intact product and Lm positive in RTE product); and
- The EIAO identifies concerns involving processes other than those originally identified for the FSA or PHRE which would require the EIAO to complete additional tools.
- The following tools are available for selection:
Table 1: Commodity-specific tools based on product type and HACCP category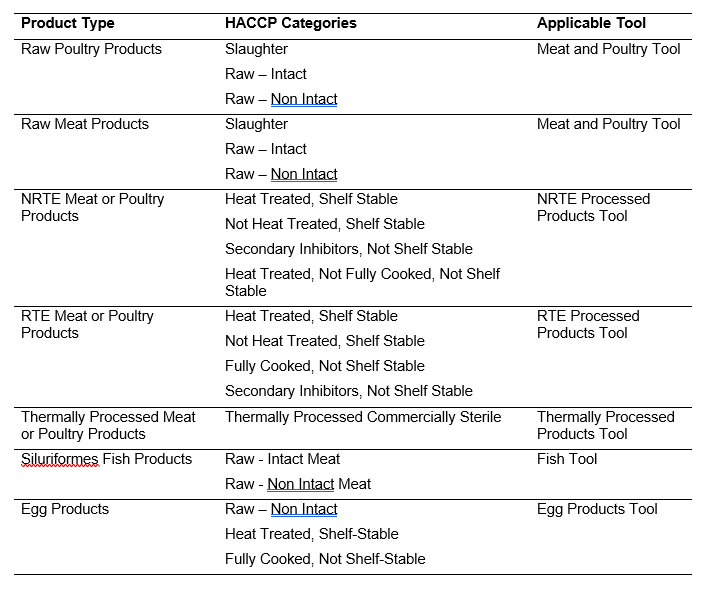
- The EIAO is to determine whether pathogen sampling, Routine risk-based Listeria monocytogenes (RLm), Intensified Verification Sampling (IVT), or other sampling is to be performed.
- Sampling is to occur prior to performing the FSA. The time for sampling is not included in the seven days allotted for the FSA.
- The EIAO is to refer to the following sampling directives for sampling and establishment notification procedures:
- RLm - FSIS Directive 10,240.5, Verification Procedures for Enforcement, Investigations and Analysis Officers (EIAOs) for the Listeria monocytogenes (Lm) Regulation and Routine Risk-Based Listeria monocytogenes (RLm) Sampling Program; and
- IVT - FSIS Directive 10,300.1, Intensified Verification Testing (IVT) Protocol for Sampling of Product, Food Contact Surfaces and Environmental Surfaces for Listeria monocytogenes (Lm) or Salmonella spp.
- The EIAO is to inventory the sampling supplies upon receipt to ensure there are enough supplies to conduct the sampling. If sampling supplies are sent to the establishment, the EIAO is to coordinate with the IPP at the establishment to inventory the sampling supplies.
- The EIAO is to arrive at the establishment the day before sampling to perform the walk-through, meet with the establishment management, and stage the sampling supplies.
- In identifying sampling sites, the EIAO is to refer to Attachment: Suggested Food Contact Surface Sites for a list of sites that have previously tested positive during RLm or IVT sampling. The EIAO is to identify additional sampling sites during the establishment tour and when meeting with IPP and establishment management.
- If the EIAO observes insanitary conditions or product adulteration at the establishment during the sampling, they are to immediately inform the IPP and the FLS, even if the EIAO has not yet started the FSA.
- The EIAO is to consider the sampling results when determining the FSA outcome. In some limited circumstances (e.g., there are unanticipated sampling delays or presumptive positives), results may not be available within the seven-day FSA timeframe. If sampling results are delayed, the DO may elect to delay the start of the FSA or delay the exit meeting until sampling results are received.
- The EIAO is to provide up to two weeks advance notice of the visit to the establishment, FLS, and IPP.
- The EIAO is to inform the establishment management of the types of documentation that need to be made available for review (e.g., last 60 production day records, HACCP plan, sampling program, sampling results). Having the documentation available at the start of the FSA will help the EIAO accomplish the FSA within the seven days.
- The DO is to manage the FSA timeline and, when necessary, utilize more than one EIAO or cross utilize EIAOs from different districts.
- ENTRANCE MEETING
- Prior to the entrance meeting with establishment management, the EIAO is to hold a pre-entrance meeting with the IIC and FLS to discuss the FSA process and answer any questions if these topics were not discussed as part of the PHRE and FSA planning process.
- The EIAO is to conduct an entrance meeting with the establishment management. The IPP and the FLS are to attend the entrance meeting, if possible. The topics to discuss during the entrance meeting include but are not limited to:
- What an FSA is;
- How it differs from the day-to-day verification activities that IPP performs;
- The reason for the FSA;
- How the scope was determined using the PHRE;
- The possible FSA outcomes;
- The EIAO's intended typical work schedule during the assessment based on the establishment's intended production schedule;
- That the EIAO may make observations during all shifts and during pre-operational activities;
- How the EIAO will access the production floor and whether any special procedures will be in place;
- Where the EIAO will conduct their work. The EIAO is to ask that they be given access to examine, copy, or scan any records that may be needed to support noncompliance determinations. If the records are stored electronically, the EIAO is ask the establishment if they would print any records that may be needed to support noncompliance determinations;
- That the EIAO will communicate findings with the IPP and establishment management as the assessment progresses;
- Whom the EIAO is to contact with questions. The plant designates various people for different processes and the EIAO is to request that the establishment identify either a telephone extension, an e-mail address, or some other way to communicate with management personnel;
- That the EIAO will confer with those establishment designees as needs for additional information arise, as well as when establishment management has questions regarding in progress FSA findings;
- That, depending on any noncompliance, the impact on food safety will determine whether NRs are given to the establishment immediately or at the exit meeting;
- That the EIAO will hold an exit meeting with establishment management at the end of the FSA;
- That the establishment will be provided a final copy of the FSA report once the FSA is complete; and
- That the EIAO will provide contact information to establishment and IPP so that they can contact the EIAO, if necessary.
- The EIAO is to document the date and the participants of the entrance meeting. The EIAO is not to document the contents of the meeting.
- INITIAL STEPS
- The EIAO is to take a tour of the establishment on the first or second day of the FSA, unless the EIAO took a tour in preparation for sampling. This tour allows the EIAO to understand the establishment's processes and flow of products and to strategize for future observations. See section VII of this chapter regarding the types of observations the EIAO is to make during the FSA.
- To best use their time during the establishment tour and the FSA, the EIAO is to:
- Prepare for the establishment tour by reviewing the flow chart and HACCP plan immediately on the first day of the FSA. After review of the flow chart and HACCP plan, the EIAO can formulate a plan to observe critical control points (CCPs), pathogen intervention applications, and possibly establishment sampling;
- Ask questions, as needed, during the tour to ensure a basic understanding of the establishment's process and flow; and
- Identify how raw and RTE areas are separated (e.g., by time, space, or separation, as well as through other means such as different colored uniforms), if performing the FSA at an RTE establishment.
- The EIAO is to start their review of the HACCP system by verifying the hazard analysis. The EIAO is to use their scientific knowledge, knowledge of Agency issuances, and professional expertise during the review. The EIAO is to assess whether the establishment has addressed hazards commonly associated with a process (9 CFR 417.5(a)(2)), and whether it can adequately support the decisions it made regarding those hazards (9 CFR 417.5(a)(1)).
- For each hazard that the establishment has determined is reasonably likely to occur, the EIAO is to verify that the HACCP plan includes one or more CCPs to control it, and that the establishment has adequate documentation to support the CCP design, critical limits, and monitoring and verification procedures as required by 9 CFR 417.5(a)(2).
- For hazards that the establishment has determined are not reasonably likely to occur because of a prerequisite program, the EIAO is to assess whether the prerequisite program supports that decision and determine whether the establishment complies with 9 CFR 417.5(a)(1) and 9 CFR 417.2(a).
NOTE: Establishments may have unique names for various prerequisite programs without incorporating “prerequisite” in the title. Temperature control programs, allergen control programs, Listeria sanitation control programs, and purchase specification programs are some examples. - If the EIAO has technical questions about the HACCP system supporting documentation, they are to discuss them with their supervisor. If questions remain, the EIAO is to submit an askFSIS question to the Office of Policy and Program Development (OPPD) as soon as possible to allow time for OPPD to research the response.
- PREREQUISITE PROGRAMS
- The EIAO is to verify prerequisite programs supporting decisions in the hazard analysis are designed and implemented effectively by evaluating whether the programs meet the following characteristics:
- The program is validated (i.e., scientific or technical support and in-plant validation data) and that the validation documents support that the hazard is not reasonably likely to occur. Section IX of this chapter discusses how to review establishment validation;
- The establishment maintains records that demonstrate that the program is being implemented as written (i.e., following and monitoring of the critical operational parameters in supporting documents) and effectively prevents the hazard (i.e., on-going verification of the decision that the hazard is not reasonably likely to occur); and
- The program describes actions that the establishment will take when it fails to implement the program or when the program has failed to prevent the hazard (i.e., corrective actions in response to an unforeseen hazard per 9 CFR 417.3(b)).
- When these characteristics are not met (e.g., systemic failure to monitor critical operational parameters), the EIAO may determine that the prerequisite program is ineffective. Since the prerequisite program is ineffective and not preventing the hazard, there is noncompliance with 9 CFR 417.5(a)(1) and 9 CFR 417.2(a). In this situation, the establishment needs to reassess its hazard analysis (9 CFR 417.4), determine whether modifications to the hazard analysis and HACCP system are necessary, and if so, make those changes to address the hazard.
- The EIAO is to verify prerequisite programs supporting decisions in the hazard analysis are designed and implemented effectively by evaluating whether the programs meet the following characteristics:
- SANITATION SOPs
Sanitation SOPs are required by regulation (9 CFR 416.12). The EIAO is to analyze and document how Sanitation SOP compliance affects the establishment's ability to support decisions in its hazard analysis or to implement its HACCP plan effectively. - SAMPLING PROGRAMS
- If sampling is part of the establishment's HACCP system (e.g., ongoing verification for a CCP or prerequisite program), the EIAO is to evaluate the written sampling procedures and the testing methods used. If the establishment conducts sampling during the FSA, the EIAO is to observe the collection procedures and document any noncompliance within the relevant tool.
- In addition to reviewing the design of the establishment's written procedures and the methods used, the EIAO is to:
- Review the sampling results for trends and determine if the process is in control. The EIAO is to review the establishment's sampling results from:
- The previous 60 days when using the Meat and Poultry tool, Fish tool, and Egg Products tool; and
- The previous 6 months when using the RTE Processed Products tool, NRTE Processed Products tool, and Thermally Processed Products tool;
- Evaluate the establishment's assessment of the results and whether the establishment completed effective and meaningful corrective actions, and if required, a reassessment, in response to sample results, if applicable; and
- Review the High Event Period (HEP) program for STEC, if applicable. The EIAO is to evaluate the criteria used to define an HEP and the support for those criteria.
- Review the sampling results for trends and determine if the process is in control. The EIAO is to review the establishment's sampling results from:
- The EIAO is to reference FSIS Directives on verifying establishment sampling including:
- FSIS Directive 10,010.3, Traceback Methodology for Escherichia coli (E. coli) O157:H7 in Raw Ground Beef Products and Bench Trim;
- FSIS Directive 10,240.4, Verification Activities for the Listeria monocytogenes (Lm) Regulation and the Ready-to-Eat (RTE) Sampling Program;
- FSIS Directive 5000.2, Review of Establishment Data by Inspection Personnel; and
- FSIS Directive 5030.1, Inspection Methodology Utilizing the Public Health Information System for the Verification of Regulatory Compliance in Egg Products Plants.
- The EIAO is to reference establishment sampling guidelines including:
- Establishment Guidance for the Selection of a Commercial or Private Microbiological Testing Laboratory;
- FSIS Compliance Guideline: Controlling Listeria monocytogenes in Post-lethality Exposed Ready-to-Eat Meat and Poultry Products;
- FSIS Compliance Guideline for Establishments Sampling Beef Trimmings for Shiga Toxin-producing Escherichia coli (STEC) Organisms or Virulence Markers;
- FSIS Guideline for Controlling Salmonella in Raw Poultry;
- FSIS Guideline for Controlling Campylobacter in Raw Poultry;
- FSIS Guideline: Modernization of Swine Slaughter Inspection - Developing Microbiological Sampling Programs in Swine Slaughter Establishments; and
- FSIS Guideline for Food Safety in Egg Products.
- The EIAO is also to review the Foodborne Pathogen Test Kits Validated by Independent Organizations database to determine whether the method used by the establishment is fit for purpose and performed under validated conditions.
- If, after reviewing these documents, the EIAO still has a question regarding the sampling program, they are to ask their supervisor or to submit a question through askFSIS.
- DIRECT OBSERVATIONS OF ESTABLISHMENT ACTIVITIES
- The EIAO is to make observations of the establishment's activities across all shifts while paying particular attention to the areas of concern identified during the PHRE. Observations provide valuable information in evaluating the establishment's food safety system. The EIAO is to make the following direct observations, conduct the following verification activities, and document findings in the relevant tools:
- The EIAO is to verify that the establishment conducts the procedures in the Sanitation SOP as written, and that the Sanitation SOP is designed effectively to prevent contamination of food contact surfaces or adulteration of products prior to operations. Since IPP routinely verify that the establishment meets the Sanitation SOP regulatory requirements (monitoring, recordkeeping, maintenance, corrective action), the EIAO is to focus their observations on evaluating whether the establishment's procedures adequately prevent cross-contamination and the creation of insanitary conditions.
- The EIAO is to verify the establishment is conducting the procedures as written in their HACCP system. The EIAO is to observe the establishment's implementation of food safety measures (e.g., CCPs, prerequisite programs) that support decisions in the hazard analysis, including antimicrobial interventions, lethality treatments, stabilization treatments, and post-lethality treatment/antimicrobial agent or process.
- During FSAs performed at slaughter establishments, the EIAO is to make direct observations of the slaughter process and sanitary dressing over multiple days, across all shifts, when available. The EIAO is to assess the sanitary dressing and process controls slaughter establishments employ in their food safety systems, considering the factors and questions presented in:
- FSIS Directive 6420.5, Verifying Poultry Slaughter Establishments Maintain Adequate Procedures for Preventing Contamination with Feces and Enteric Pathogens;
- FSIS PHIS Directive 6410.1, Verifying Sanitary Dressing and Process Control Procedures in Slaughter Operations of Cattle of Any Age;
- FSIS Directive 6410.4, Verifying Swine Slaughter Inspection System: Ante-Mortem and Post-Mortem Inspection and Verification of Food Safety and Ready-to-Cook Requirement;
- FSIS Guideline for Controlling Salmonella in Raw Poultry;
- FSIS Guideline for Controlling Campylobacter in Raw Poultry;
- Industry Guideline for Minimizing the Risk of Shiga Toxin-Producing Escherichia coli (STEC) in Beef (including Veal) Slaughter Operations;
- FSIS Guideline to Control Salmonella in Swine Slaughter and Pork Processing Establishments;
- Guideline for Training Establishment Sorters under the New Swine Slaughter Inspection System; and
- Compliance Guideline for Training Establishment Carcass Sorters in the New Poultry Inspection System (NPIS).
- The EIAO is to make direct observations of the establishment's processing activities. The EIAO is to assess the process controls the establishment employs in their food safety systems, considering the guidance presented in:
- Industry Guideline for Minimizing the Risk of Shiga Toxin-Producing Escherichia coli (STEC) in Beef (including Veal) at Processing Operations;
- FSIS Guideline for Controlling Salmonella in Raw Poultry;
- FSIS Guideline for Controlling Campylobacter in Raw Poultry;
- FSIS Guideline for Food Safety in Egg Products;
- FSIS Guideline to Control Salmonella in Swine Slaughter and Pork Processing Establishments;
- FSIS Cooking Guideline for Meat and Poultry Products (Revised Appendix A); and
- FSIS Stabilization Guideline for Meat and Poultry Products (Revised Appendix B).
- The EIAO is to make direct observations of any establishment sampling, and if applicable, the establishment's in-house laboratory, to ensure the establishment is following the procedures in its written program.
- The EIAO is to make observations of the establishment's activities across all shifts while paying particular attention to the areas of concern identified during the PHRE. Observations provide valuable information in evaluating the establishment's food safety system. The EIAO is to make the following direct observations, conduct the following verification activities, and document findings in the relevant tools:
- RECORDS REVIEW
- The EIAO is to review documents associated with the HACCP system. The EIAO is to prioritize records necessary to answer questions in the FSA tools and to evaluate whether the establishment is maintaining an adequate food safety system. These will include, but are not limited to, records directly relevant to:
- Sanitary dressing;
- Prerequisite programs;
- Establishment interventions;
- Lethality and stabilization procedures;
- Establishment sampling results; and
- Effectiveness of corrective actions.
- The EIAO is to randomly select 13 production days from the preceding 60 days and to review data from those 13 days. The EIAO is not to review each day's records from the preceding 60 days. This limited review will provide the EIAO with knowledge of how the HACCP system design is implemented and whether it meets regulatory requirements.
- If an establishment has operated for fewer than 13 days in the preceding 60 days, the EIAO is to review data that goes back further than 60 days, until they have reviewed 13 days of data.
- The EIAO is also to review more than 13 days of records if the record review indicates a larger food safety concern (e.g., the EIAO identifies numerous deviations the establishment did not identify).
- The EIAO is to assess the recordkeeping system design and implementation, including whether the results of the monitoring and on-going verification procedures appropriately reflect the implementation of the establishment's HACCP system. The EIAO is not to focus on compliance with basic recordkeeping requirements (e.g., signature and dating requirements in 9 CFR 417.5(b)), as IPP verify those requirements of individual records. If there is a systemic problem with basic recordkeeping requirements, the EIAO is to notify the FLS.
- The EIAO is also to review the records to determine whether there were any deviations from the establishment's critical limits that were not detected by the establishment's monitoring procedures.
- The EIAO is to evaluate whether the in-plant team receives the appropriate sampling tasks based on the products produced and the sampling results in the PHRE PHIS report. If the EIAO identifies that the appropriate sampling tasks are not being assigned to the in-plant team, they are to contact the FLS.
- To determine sampling program eligibility the EIAO is to review the following issuances:
- FSIS Directive 10,010.1, Sampling Verification Activities for Shiga Toxin-producing Escherichia coli (STEC) in Raw Beef Products;
- FSIS Directive 10,240.4, Verification Activities for the Listeria monocytogenes (Lm) Regulation and the Ready-to-Eat (RTE) Sampling Program;
- FSIS Directive 10,250.1, Salmonella and Campylobacter Verification Program for Raw Poultry Products;
- FSIS Directive 10,800.1, Residue Sampling, Testing and Other Verification Procedures under the National Residue Program for Meat and Poultry Products;
- FSIS Directive 10,230.3, FSIS Verification Testing of Domestic Egg Products; and
- Current FSIS Sampling Notices.
- The EIAO is to review documents associated with the HACCP system. The EIAO is to prioritize records necessary to answer questions in the FSA tools and to evaluate whether the establishment is maintaining an adequate food safety system. These will include, but are not limited to, records directly relevant to:
- VALIDATION DOCUMENTS
- The EIAO is to review the two types of supporting documentation required under 9 CFR 417.4(a)(1) to determine whether the HACCP system is validated:
- Adequate scientific or technical support for the HACCP system design (design); and
- In-plant validation data (execution) demonstrating the establishment can implement its system as designed.
- The EIAO is to review the FSIS Compliance Guideline: HACCP Systems Validation that includes recommendations for meeting the validation requirements.
- To determine whether the establishment maintains adequate scientific support for the design of its HACCP plan (including CCPs and critical limits) or prerequisite program (e.g., Sanitation SOPs, equipment specifications), the EIAO is to evaluate whether:
- The establishment maintains documentation of the scientific and technical support for the design of its HACCP system;
- The scientific support is complete and contains the methodology and results;
- The methodology is appropriate for the purpose;
- The results in the scientific support demonstrate that the establishment's process prevents, reduces, or eliminates the hazard to acceptable levels;
- The scientific and technical support closely relates to the establishment's actual process, product, and hazard identified in the hazard analysis. If it does not closely relate, the EIAO is to evaluate whether the establishment has support or justification (science-based rationale) for why the scientific support should still apply to its process; and
- The establishment incorporates the same critical operating parameters described in the scientific and technical support into its CCPs, prerequisite programs, and other programs. Processes or programs that do not incorporate the same critical operating parameters as the scientific or technical support require additional support (science-based rationale) for those changes. The EIAO is to evaluate the additional support to ensure it is adequate.
- To determine whether the establishment maintains adequate in-plant validation data demonstrating that their HACCP system works as intended, the EIAO is to evaluate whether:
- The establishment collected and maintained in-plant validation data for at least one product from each HACCP processing category;
- The in-plant validation data consists of data demonstrating that the critical operational parameters of the process are being met. If the establishment has adequate scientific or technical support and is following the parameters in the scientific or technical support, then in-plant microbiological data is not needed to comply with the initial validation requirements. When the establishment does not have adequate scientific or technical support, or when it is not following the parameters in the scientific or technical support, the EIAO is to evaluate whether the in-plant validation data also consists of microbiological data. If the in-plant validation data does not consist of microbiological data, the EIAO is to assess how this impacts the establishment's food safety system.
- The establishment collected in-plant validation data from 90 calendar days. For large establishments, 90 calendar days equates to approximately 60 production days. For small and very small establishments, 90 calendar days equates to a minimum of 13 production days;
- The data reflects the process as currently designed; and
- The establishment reviewed the in-plant validation data during the initial validation period to determine whether it supports that the system can be implemented as designed.
- The EIAO is to document noncompliance if the establishment has inadequate support.
- The EIAO is to review the two types of supporting documentation required under 9 CFR 417.4(a)(1) to determine whether the HACCP system is validated:
- ANSWERING FSA TOOL QUESTIONS
- The EIAO is to document relevant vulnerabilities or noncompliance findings in the FSA tools.
- The EIAO is to limit answers in the FSA tool to the question being asked, the HACCP category or categories being evaluated, and any vulnerabilities or noncompliance identified. The EIAO is not to include information from other HACCP processing categories unless the information has a bearing on the HACCP category being evaluated as a part of the FSA. Limiting responses allows the EIAO to focus their time towards building a supportable recommendation.
- When responding to the questions in the tool, the EIAO is to keep in mind that several questions could have similar responses, depending on the issues at the establishment. In these situations, the EIAO is to “cut and paste” their responses from the previous question(s).
- The EIAO is to complete the Decision-Making Analysis, FSA recommendation (section XII of this chapter), and Executive Summary (section XI of this chapter) questions in the General tool after all other questions are answered.
- The EIAO is to use the Decision-Making Analysis question as context and support for the recommendation. The EIAO is to use sampling results (RLm, IVT), PHRE, in-plant observations, and the HACCP system design and implementation findings documented in the tools as support for recommendations. The EIAO is to also discuss and interpret their major findings and how the findings impact the establishment's ability to produce safe, wholesome, and unadulterated product in no more than two pages.
- EXECUTIVE SUMMARY
- The EIAO is to write the Executive Summary as a brief overview (less than 350 words) of the FSA recommendation, the major findings (generally three to five key findings), and essential information that support that recommendation. The EIAO is to include additional information in the Decision-Making Analysis question.
- The EIAO is to use the information in the summary question from each tool to write the Executive Summary.
- The EIAO is not to duplicate the Decision-Making Analysis Section.
- The EIAO is to write the Executive Summary after completing the Decision-Making Analysis and the FSA recommendation questions.
- The EIAO is to write the Executive Summary to stand alone in explaining and supporting the recommendation without reference to any other part of the FSA. An Executive Summary is to contain the following:
- One sentence that describes the EIAO's recommendations;
- One or two sentences that describe the establishment, its processes, and the major types of products it produces;
- One or two sentences that describe the establishment's compliance history;
- One sentence that describes the sampling results, if applicable;
- One or two sentences that describe the major findings leading to the recommendation; and
- One or two sentences that discuss the significance of those regulatory findings that result in the establishment not meeting the requirements of the Acts, and the effect those findings have on the establishment's ability to produce safe products, if applicable.
- The EIAO is not to introduce any new information into the Executive Summary that is not captured in the FSA tools.
- FSA RECOMMENDATIONS
- The EIAO is to recommend one of the three following outcomes for the FSA: No Further Action, Issuance of NRs, or Enforcement Action.
- No Further Action
The EIAO is to describe the support for how the establishment meets the applicable regulatory requirements and that no food safety concerns exist. - Issuance of NRs
- The EIAO is to ensure the information for NRs is consistent with FSIS Directive 5000.1, Verifying an Establishment's Food Safety System.
- The FLS is to ensure that IPP input the NRs appropriately (including the appropriate regulatory citation) and issue the NRs during the exit meeting, if not already provided earlier. If the NR(s) were completed as part of the FSA, IPP are to include that information in the description of the NR(s) and check the designated box in PHIS.
- The FLS is to conduct an in-person follow-up visit within 30 days of the exit meeting when the EIAO identifies systemic issues during the FSA that are documented by IPP in NRs. During this and any additional visits, the FLS will verify the establishment's response provided to address the noncompliance. The establishment's response will include specific corrective actions for certain regulations, such as when there is a Sanitation SOP noncompliance involving direct product contamination.
- The FLS is to document findings during the follow-up verification visits in a Memorandum of Interview according to FSIS Directive 5010.1, Food Safety Related Topics for Discussion During Weekly Meetings with Establishment Management. The FLS is to describe in detail the establishment's progress in implementing its response including any corrective and preventive actions and any additional information, as appropriate.
- The FLS may need to conduct additional in-person follow-up visits depending on the establishment's response to address the noncompliance and any corrective or preventive measures. For example, if the establishment replaces equipment due to Lm harborage that could not be eliminated or implements a new critical control point requiring it to gather in-plant validation data over 90 calendar days, the FLS may conduct additional in-person follow-up visits to verify those actions are effective. The FLS is to correlate with IPP to consider observations, information, and noncompliances IPP have gathered since the FSA exit meeting and to look for any patterns or trends in the findings. The FLS is to contact the EIAO if there are questions about the establishment's response to the NR(s).
- The EIAO is to work with the DO and FLS to determine if any further FSIS follow-up action is required once the establishment has implemented corrective actions, if applicable. The EIAO is to contact the FLS within 30 days of the exit meeting to determine the status of NR(s) and follow-up visit findings.
- Enforcement Action
- Enforcement action includes an NOS or an NOIE, with or without NRs. The NOIE will include NRs if the EIAO observes noncompliances during the FSA. The NOIE will not include NRs if IPP observe the noncompliance, either before or during the FSA. The DO issues the NOS or NOIE.
- If the EIAO observes conditions in the establishment that supports taking a withholding or suspension action without prior notification (9 CFR 500.3), the EIAO is to first stop the wrongful practice. The EIAO is to contact the DO to determine how to proceed. The EIAO is to ensure the responses in the FSA tools support the recommendation.
- If the EIAO recommends an NOIE, the responses in the FSA tools must document the establishment conditions that support the NOIE (e.g., HACCP plan is inadequate, Sanitation SOP is not properly maintained or implemented, establishment has not maintained sanitary conditions) (9 CFR 500.4). The EIAO is to refer to FSIS Directive 5100.3 for more information on NOIEs.
- The EIAO is to collect, safeguard, and transfer evidence as described in FSIS Directive 8010.3, Procedures for Evidence Collection, Safeguarding and Disposal.
- The EIAO may also collaborate with the DCS in creating and maintaining case files in Assurance Net, as described in FSIS Directive 5100.3, Administrative Enforcement Reporting (AER) System.
- FSA FINALIZATION
The DO is to manage the review process for FSAs. The SEIAO and DCS are to review FSAs that result in a recommendation of enforcement. The DO is not to review responses to all questions within the FSAs. Rather, the DO is to use their discretion in reviewing responses according to the public health risk. - EXIT MEETING
- Once the DO has reviewed the FSA, the EIAO is to schedule the exit meeting with the establishment management. The IPP and FLS are to attend the exit meeting, if possible, but the EIAO is not to delay the exit meeting to accommodate their attendance. The exit meeting is to take place on the last day of the FSA, within the seven days. The EIAO is to document the date of the exit meeting and the attendees in the General tool.
- During the exit meeting, the EIAO is to:
- Thank the establishment for its cooperation;
- Describe the FSA findings, including any recommendations the EIAO made to the DO;
- Describe the basis for all NRs and any enforcement recommendations. If the DO issues an enforcement action, the EIAO is to give the NOIE or NOS at the exit meeting;
- Provide a draft or final copy of the FSA to the establishment management. If a draft copy is provided, send a final copy once the exit meeting information has been added; and
- Answer any questions from the establishment.
- The EIAO is to direct the small and very small establishments to FSIS guidance resources.
- QUESTIONS
Refer questions regarding this directive to your supervisor and if needed to the Office of Policy and Program Development through askFSIS or by telephone at 1-800-233-3935. When submitting a question, complete the askFSIS and select HACCP Deviation & HACCP Validation for the inquiry type.
NOTE: Refer to FSIS Directive 5620.1, Using askFSIS, for additional information on submitting questions.
Attachment. Suggested Food Contact Surface Sites
Purpose: This document is designed to assist the EIAO in choosing a food contact sampling site for RLm and IVT sampling.
Historical positive results from RLm and IVT testing conducted by FSIS have been used to summarize the most common Food Contact Surface (FCS) positive sites. The EIAO is to use the following list as a foundation for planning RLm sampling prior to beginning an FSA. The final sampling plan should additionally reflect historical areas of concern at the establishment and areas of concern identified by the IPP and FLS.
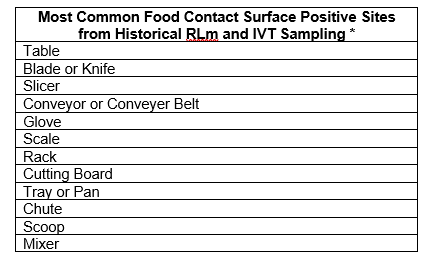
* Additional discussion of FCS sites can be found in the FSIS Compliance Guideline: Controlling Listeria monocytogenes (Lm) in Post-lethality Exposed Ready-to-Eat (RTE) Meat and Poultry Products.
