
Raw Pork Products Sampling Program
I. PURPOSE
A. This notice is being reissued to inform inspection program personnel (IPP) that sampling under the Raw Pork Products Sampling Program will continue. All raw pork samples will continue to be analyzed for Salmonella and for aerobic counts (AC), which serve as indicator organisms. These data will be used for policy development, which will include developing new performance standards for Salmonella in raw comminuted pork and intact or non-intact pork cuts. The project codes and sampling instructions to IPP at establishments that produce raw pork products remain unchanged.
B. Significant changes in this revision include a separate section defining product eligibility;
information to assist IPP in better scheduling sampling to ensure lab capacity; instructions to perform the establishment profile task to ensure correct sampling assignments; definition of box-in-box-out product eligibility; and updated instructions and links to the new askFSIS application.
II. BACKGROUND
A. As explained in the January 26, 2015, Federal Register Notice, Changes to the Salmonella and Campylobacter Verification Testing Program: Proposed Performance Standards for Salmonella and Campylobacter in Not-Ready-to-Eat Comminuted Chicken and Turkey Products and Raw Chicken Parts and Related Agency Verification Procedures and Other Changes to Agency Sampling (80 FR 3940), FSIS implemented exploratory sampling of raw pork products to test for pathogens of public health concern, as well as for indicator organisms in May 2015.
B. The Agency continued to collect and analyze raw pork products in an interim phase before implementing Phase II of the Raw Pork Product Exploratory Sampling Program. In Phase II, sampling focused on Salmonella and indicators in both slaughter and processing establishments and Shiga toxin-producing E. coli (STEC) in slaughter establishments only. A summary of the sample results can be viewed online at: Quarterly Sampling Results for FSIS-Regulated Products, and further analysis of the data can be viewed online at: Nationwide Raw Pork Products Exploratory Sampling Program.
C. The Agency will continue to analyze raw pork product samples for Salmonella and indicator organisms. The sampling data will be used to develop future policies designed to reduce the levels of Salmonella in raw pork products.
III. ESTABLISHMENT SAMPLING ELIGIBILTY AND PUBLIC HEALTH INFORMATION SYSTEM (PHIS) SAMPLING TASK ASSIGNMENT
A. IPP will continue to see the raw pork sampling tasks appear in PHIS for eligible establishments. The number of tasks per month are assigned as shown in Table 1, as determined by the establishment’s production volume in its PHIS profile.
Table 1: Raw Pork Products Sampling Eligibility
B. FSIS will schedule five (5) samples per month for eligible establishments producing greater than 6,000 pounds of comminuted product and five (5) samples per month for those eligible establishments producing greater than 50,000 pounds per day of pork cuts (intact and non-intact).
C. IPP are to be aware that eligible establishments will receive sampling tasks based on the establishment’s average daily volume (pounds per day) and the number of days of production that are entered into the PHIS establishment profile for the eligible product groups listed in Table 2 and Table 3 of this notice. IPP are also to be aware that establishments producing 1,001 to 6,000 pounds of comminuted pork products or 1,001 to 50,000 pounds of pork cuts will be randomly selected for sampling tasks.
D. IPP are to schedule PHIS tasks as follows:
- Schedule the samples on the PHIS calendar on days that the establishment indicates that the product will be produced; and
- When possible, schedule at a frequency of no more than once per week. At eligible establishments that receive 5 tasks per month, IPP are to schedule more than one sample to ensure that all 5 samples are collected each month.
E. IPP are to perform the monthly PHIS Update Establishment Profile Task following FSIS Directive 5,300.1, Managing the Establishment Profile in the PHIS, ensuring that the product is correctly documented in the profile, including Hazard Analysis Critical Control Point (HACCP) processing category, finished product category, and production volume category. IPP are to be aware that:
- Instructions on how to calculate the production volume information are listed in FSIS Directive 5,300.1, Section VIII, Production Volume Information;
- If the product is box in-box out, meaning product is stored at the establishment without re-portioning, it should not be included in the finished product category information and is ineligible for sampling; and
- Further guidance to ensure eligible products are correctly entered into the PHIS profile is provided in IPP Help.
F. IPP are to notify official establishment management just before collecting a sample. Establishments are not required to hold the sampled production lot pending the sample results.
G. Descriptions and examples of products not eligible for sampling are available in Table 4. IPP are not to collect samples of ineligible products.
Table 2. Eligible Raw-Intact and Non-Intact Pork Cuts Finished Product Category: Raw-intact and Raw, non-intact pork
Table 3. Eligible Raw Comminuted Products Finished Product Category: Raw ground, comminuted, or otherwise non-intact pork
Table 4: INELIGIBLE PRODUCTS – DO NOT SAMPLE
H. IPP Help has a menu item, Sampling, that houses the Raw Pork menu. The Raw Pork menu contains sample collection instructions, in-depth product descriptions, and product examples of the PHIS pork product groups to help IPP update the establishment profile. IPP can access IPP Help by double-clicking the FSIS Applications icon from the desktop of their government-issued computer.
I. IPP are to refer to FSIS Directive 13,000.2, Performing Sampling Tasks in Official Establishments Using the Public Health Information System, for instructions on how to add the task to the Task Calendar, enter the sample information, submit the sample information to the laboratory, and print a finalized sample collection form in PHIS.
J. IPP are to complete all requested sample information and complete any sample questionnaires in PHIS. The list of questions for each sampling task is included in IPP Help. IPP are to ensure that all questions in the PHIS questionnaire are answered accurately.
K. IPP are to collect and submit the sample to the FSIS laboratory within the sample collection window assigned to the sampling task.
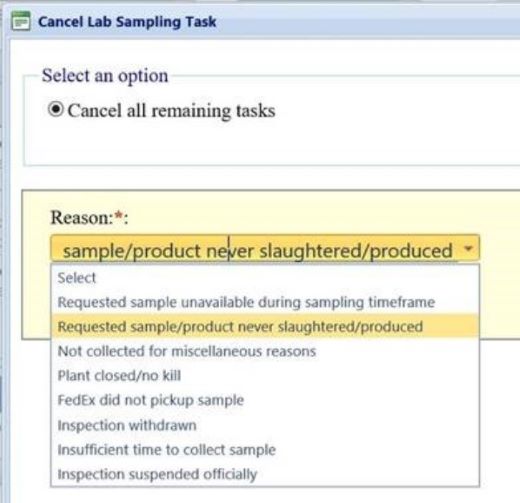
L. If sampling tasks remain in the task list at the end of the sampling window, IPP are to cancel them from the task list and provide the correct reason, as shown in the image below. If none of the listed reasons are appropriate, IPP are to select Not collected for miscellaneous reasons and provide additional details in the text box provided.

M. If an establishment does not produce eligible product, IPP are to cancel any remaining product verification sampling tasks. IPP are to cancel these tasks from both the “Establishment Task List” (Delete this task from the Task List) and, if scheduled, the Task Calendar, using the correct option (i.e., cancel this task and remove it and all other instances of this task from the Task List). In addition, IPP are to provide the reason for canceling the task, Requested sample/product never slaughtered/produced, as shown in the image below.
N. IPP are also to update the PHIS Establishment Profile accordingly if the profile has inaccurate or incomplete information about the products the establishment produces.
NOTE: Although the FSIS Directive 5,300.1, states that IPP can wait until the next monthly Establishment Profile Inspection Task to make updates to the “Establishment Profile,” the profile entry impacts sampling eligibility. Therefore, when IPP become aware of incorrect information regarding establishment product within the PHIS Establishment Profile, IPP are to immediately correct and update the known product changes in PHIS. IPP are to select “0” from the drop-down list for “Average Daily Volume” when IPP become aware the establishment has temporarily stopped producing a product. IPP are to delete products from the profile during the same shift they become aware the establishment has indefinitely stopped producing that product. IPP are to be aware updates to the profile will not impact sampling tasks already assigned in PHIS, and IPP will need to continue to cancel those tasks.
IV. SAMPLE SELECTION AND ELIGIBILITY CRITERIA
A. IPP are to refer to the PHIS sampling tasks for information on the product to collect. At establishments producing more than one eligible product group, IPP may be assigned more than one sampling task with more than one sample project code during the same sampling window.
B. IPP are to use a method for randomly selecting the production lot for sampling and use the same method each time IPP in the same establishment perform sample collection. IPP are to randomly select a day, shift, and time within the sampling window shown in PHIS. IPP are to collect samples from all shifts that the establishment operates. When possible, IPP are to collect samples under each project code at a frequency of no more than once per week, with sample collection spread out randomly and evenly throughout the month. In the case that there are more samples than weeks in the sampling window, IPP are to schedule more than one sample in a given week.
C. The following practices do not exclude products from routine sampling:
- Addition of ingredients such as spices, seasonings, rosemary extracts, or fruits/vegetables to eligible pork products;
- Application of an antimicrobial treatment or intervention (other than a treatment that achieves full lethality);
- Addition of meat or poultry products from a different species to eligible pork products; and
- Sending product to hotels, restaurants, or similar institutions (HRI).
D. The following products are not to be included in routine sampling:
- Battered or breaded pork product, such as dumplings, egg rolls, or pot stickers;
- Products in the HACCP processing category “Heat-treated but not Fully Cooked - Not Shelf Stable,” such as partially cooked products or products that contain cooked pork and raw dough such as pot pies; and
- Raw pork products intended for use in ready-to-eat (RTE) products at the establishment or another official federally inspected establishment.
E. FSIS does not sample product that will receive full lethality treatment at a federally inspected establishment provided that the establishment’s hazard analysis and flow chart show that the product is intended for such use.
F. If the establishment meets the requirements in D.3 and E. of this section, IPP are to verify during the performance of the associated HACCP verification task that the intended use of all the product the establishment produces is for processing into RTE product (9 CFR 417.2(a)(2)). IPP are to verify that all product will be processed into RTE product by:
- Observing that all the product moves to be further processed into RTE product in the establishment;
- Reviewing records to ensure that all products are further processed into RTE products in the establishment. Records may include those containing production codes or production lot codes; or
- Reviewing the establishment’s HACCP plan and hazard analysis for the intended use of the products and verifying that the establishment has procedures incorporated in its food safety system that document the movement of all product from that product class to another official establishment at which the product is further processed into RTE product.
V. RAW INTACT AND NON-INTACT PORK CUTS
A. Table 2 identifies which product groups and products are eligible for sample collection under the HC_PK_CUT01 sampling task.
B. IPP are to collect fresh, not frozen, raw pork samples in final packaging whenever possible with an appropriate number of packages to equal 2 lbs. ± 10%. IPP are to place the product collected in its final packaging into the larger, non-sterile bag provided with the sampling supplies.
C. For intact and non-intact pork cuts that are not available in their final packaging, IPP are to use the single larger Whirl-Pak bag and aseptically collect one or more cuts to fill the Whirl-Pak bag leaving 2 to 3 inches of space at the top of the bag. IPP are to expel as much air as possible from the bag before closing.
NOTE: IPP are not to use the Whirl-Pak bags when collecting products in final packaging.
D. For establishments with intact and non-intact pork cuts that are processed in a frozen state only (e.g., all product is received frozen and processed frozen; product further processed in frozen state only), IPP are to collect a frozen 2 lbs. sample if the establishment does not produce any other eligible pork product. IPP are to indicate whether the sample is fresh or frozen in the PHIS questionnaire.
E. Individual whole pork cuts (intact or non-intact) larger than 2 lbs. are not to be collected by IPP.
F. IPP are to access the IPP Help menu item, Sampling, then Raw Pork Sampling Program Guidance, for more detailed information for collecting raw pork samples.
VI. COMMINUTED PORK PRODUCTS
A. Table 3 identifies which product groups and products are eligible for sample collection under the HC_PK_COM01 sampling task.
B. IPP are to collect fresh, not frozen, comminuted pork product samples in their final packaging, whenever possible, and an appropriate number of packages to equal 2 lbs (+/- 10%). IPP are to place the product collected in its final packaging into the larger, non-sterile bag provided with the sampling supplies.
C. For all comminuted pork product samples not available in their final packaging, IPP are to use the two (1 pound each) Whirl-Pak bags when collecting aseptic grab samples. The Whirl-Pak bags have fill lines to help IPP collect the desired sample weight of 2 pounds (i.e., 1 pound in each of the two Whirl-Pak bags). IPP are to collect enough product to fill each of the two Whirl-Pak bags to the fill-line.
NOTE: IPP are not to use the Whirl-Pak bags when collecting products in final packaging.
D. For comminuted pork products that are processed in a frozen state only (e.g., all product received is frozen and processed frozen; product further processed in frozen state only), IPP are to collect a frozen 2-lb. sample if the establishment does not produce any other eligible pork product. IPP are to indicate whether the sample is fresh or frozen in the PHIS questionnaire.
E. For mechanically separated or AMR (finely textured pork) product samples, IPP are to fill each of the two Whirl-Pak bags by using the sanitized scoop and spatula contained in the enhanced sampling supplies. When needed, IPP are to request enhanced sampling supplies via Outlook email at least 3 business days before the scheduled day of sampling. The enhanced sampling supplies will contain all HC_PK_COM01 supplies plus: one 4-ounce sterile scoop, one sterile spatula, and two towelettes.
F. To request enhanced sampling supplies via Outlook email, IPP are to select one of the following addresses:
SamplingSupplies-EasternLab@usda.gov
SamplingSupplies-MidwesternLab@usda.gov
SamplingSupplies-WesternLab@usda.gov
G. IPP are to use the e-mail subject heading “Enhanced Raw Pork Products Sampling Program sampling supplies.” IPP are to request one set of enhanced sampling supplies for each sample collection event. IPP are to include in their request the establishment name and street address (no P.O. Box).
H. IPP are to access the IPP Help menu item, Raw Pork Sampling Program Guidance, for more detailed information on collecting samples of comminuted pork products and the enhanced sampling supplies.
VII. SAMPLING SUPPLIES FOR RAW PORK PRODUCTS SAMPLING PROGRAM
A. The FSIS laboratories will ship the sampling supplies automatically to the IIC at the establishments selected for sampling. Supplies will arrive in a shipping container labeled with either the HC_PK_COM (comminuted products) or the HC_PK_CUT (cuts) sorting label. IPP are to refer to IPP Help for a list of sampling supplies that they should receive. IPP are to use only the sampling supplies provided by the laboratories. Sampling boxes and supplies from the exploratory sampling project (EXP_PK_COM02, EXP_PK_ICT02, EXP_PK_NCT02) may be used to collect samples under the verification sampling program (HC_PK_COM01 and HC_PK_CUT01), as the supplies have not changed. Shipping boxes will be relabeled as they are repacked by the receiving laboratories.
B. If IPP do not receive sampling supplies by the first day of the collection window or need to request replacements for damaged or lost supplies, they are to request Raw Pork Product Sampling Program sampling supplies. For example, if raw pork products sampling tasks are assigned in PHIS in late October, IPP are to request replacement supplies only if they have not arrived by November 1. IPP requiring sampling supplies are to request them through PHIS or Outlook email 3 business days (not including weekends and holidays) before they intend to collect the sample.
- To request sampling supplies via PHIS, IPP are to right-click a scheduled lab sampling task (e.g., HC_PK_COM01, HC_PK_CUT01) on the Task Calendar, then select Order Supplies from the drop-down menu.
- To request sampling supplies via Outlook email, IPP are to select one of the following addresses:
SamplingSupplies-EasternLab@usda.gov
SamplingSupplies-MidwesternLab@usda.gov
SamplingSupplies-WesternLab@usda.gov
C. IPP are to use the subject heading “Raw Pork Product Sampling Program Sampling Supplies” in the email and include the establishment name and number, the project code (HC_PK_COM01, HC_PK_CUT01) the IPP’s contact name, establishment number, telephone number, and a list of the supplies needed.
D. IPP are to return any unused shipping containers and sampling supplies for these projects to the FSIS Field Service Laboratories. IPP are to send a request for a pre-addressed return FedEx ground shipping label to one of the email addresses listed above.
E. IPP are to access the IPP Help menu item, Raw Pork Sampling Program Guidance, for more detailed information on collecting samples of comminuted pork products and the enhanced sampling supplies.
F. IPP are to refer to IPP Help, Requesting Sampling Tasks, Discards, or Modifications, for further help requesting additional sampling, discards, or modifications to sampling tasks.
VIII. RESULTS
Salmonella results will be posted in LIMS-Direct as soon as results are available. Individual sample results will not result in regulatory control actions. Therefore, establishments are not required to hold the sampled production lot pending the Salmonella sample results. However, repetitive positive sample results over time may indicate a concern with respect to process control and HACCP system support. IPP are to seek guidance from their supervisor for further instruction regarding these or other concerns.
IX. DATA ANALYSIS
The FSIS Office of Public Health Science (OPHS) and Office of Planning, Analysis, and Risk Management (OPARM) will analyze the data collected in the Raw Pork Product Sampling Program to monitor Salmonella in raw products.
X. QUESTIONS
Refer questions regarding this notice to your supervisor or as needed to the Office of Policy and Program Development through askFSIS or by telephone at 1-800-233-3935. When submitting a question, complete the web form and select Sampling as the Inquiry Type.
NOTE: Refer to FSIS Directive 5,620.1, Using askFSIS, for additional information on submitting questions.
See PDF for additional information
